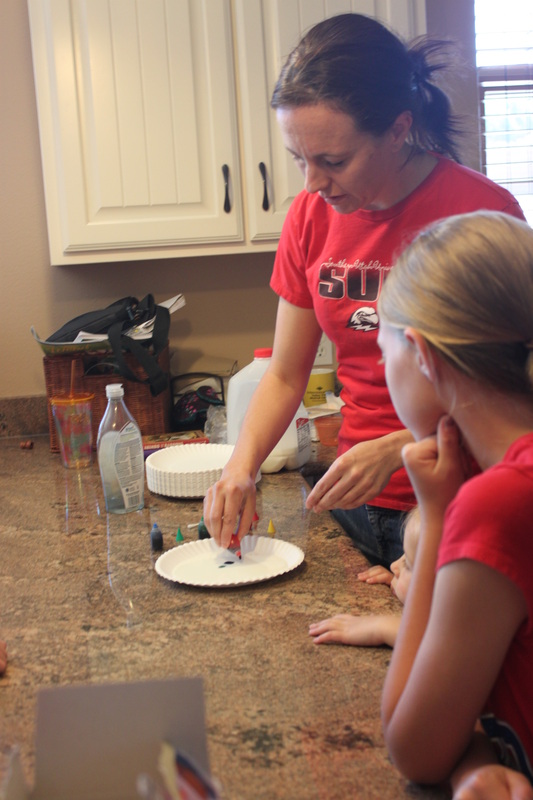Our Backyard Book Club pick this month was Fortunately, the Milk by Neil Gaiman, so naturally, we needed to do a milk science activity. This is a classic, colorful, and very cool “trick” that appeals to lots of ages. But of course, it’s even cooler if you know the science behind it.
Materials: milk, dish soap, cotton swabs, food coloring, plates
What is milk made of? Calcium and vitamins were the first two ingredients our kids named, and most of them knew there was fat in there. But some were surprised to learn that milk is mostly made of water. We learned a little about hydrophobic (molecules that are “afraid” of water) and hydrophilic (molecules that “love” water). But we also talked about how molecules just react and respond to their surroundings, and they don’t actually have feelings like fear and love. 🙂
Fats and oils are hydrophobic, but in milk, the fat is just suspended. That means it’s mixed throughout, but not really dissolved. And it’s not totally “happy” about it. So what would happen if we gave it another option? If it didn’t have to be all mixed up with the water?
That’s where the dish soap comes in. Dish soap molecules have two parts hooked together–a hydrophobic side and a hydrophilic side. So they’ll dissolve in water, but they’ll also be attractive to hydrophobic molecules like fats and oils. (That’s why it can get those things off your dishes! For a more in-depth explanation, check out this article.) If you mix dish soap with milk, those fat molecules will race to hook up with the hydrophobic ends of the soap molecules. And that race is pretty fun to watch.
Step 1: Pour just enough milk onto the plate to cover the bottom.
Step 2: Put one drop of each food coloring near the center of the plate, but not touching.
Step 3: Cover the end of a cotton swab with dish soap.
Step 4: Dip the cotton swab into the center of your plate, between the drops of food coloring and watch closely!
Step 5: Experiment! You can hold the cotton swab still, or gently twist it back and forth, or dip it on other parts of your plate. You can put the food coloring on different parts of the plate and try to drive them to the center. You can use different kinds of milk (whole, skim, etc.) and see what difference it makes. So beautiful! So fun!